|
4/15/2021 0 Comments Ferrous Vs Ferric Mnemonic
Iron is an element in the d block of the periodic table, which means it is a transition metal.These metals have a special characteristic of exhibiting multiple valencies.
Therefore, transition metals can have several stable oxidation states. 
The main difference between Ferrous and Ferric is that Ferrous is the2 oxidation state of Iron while Ferric is the 3 oxidation state of Iron. Its oxide is green in color, and it is the first step in the formation of rust. As Iron is a metal, it needs to release or give away electrons and form positive ions, in order to gain a stable electron configuration. The general electron configuration for the iron element can be written as 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 6. When it comes to ferrous, two electrons are released from the 4s orbitals as it is the outermost orbital. However, 3d orbitals are higher in energy than the 4s orbitals. Therefore, discharge of electrons takes place from the 4s orbitals, even though electrons are filled into the 3d orbitals at last. Now this ionic form is able to form compounds and complexes with other elements and negatively charged ions. These complexes can also be in ionic form and, in that case, they are generally known as transition metal compounds. When naming this kind of inorganic complexes, the term Ferrous is often replaced with the names of the element and its valency is given within brackets, such as Iron (ll) oxide. Some examples for famous ferrous compounds are FeO, FeCl 2, FeSO 4, etc. Upon the oxidation of Iron, it turns into ferrous oxide and in the presence of more oxygen the ferrous ion get oxidized to form ferric ions, in this case ferric oxide which is rust. 
The resulting electron configuration of the ferric ion would be: 1s 2 2s 2 2p 6 3s 2 3p 6 3d 5. This configuration has more stability than the ferrous ion and also the elemental form. As this configuration satisfies the partial electron filling. Any configuration where the orbitals are fully-filled or half-filled is considered to be higher in stability than the other forms. In the case of ferric ion, each d orbital contains an electron, making it half-filled as d orbitals contain five electrons. Ferric ions too form other compounds and complexes, and when naming inorganic complexes, it is written as Iron (lll). 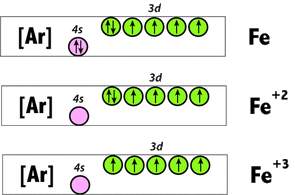
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
